
Why Lead Batteries Are Important for the Shift to EV Ecosystems
Leave a CommentAs automobiles evolve, so does the role of the battery. Automakers, regulators, and consumers require more power, safety, and sustainability from automotive batteries, especially electric vehicle (EV) batteries. To meet these growing demands, absorbent glass mat (AGM) advanced lead batteries are ideal for 12-volt systems to facilitate the global shift toward autonomous and electric vehicles. […]

Clearing the Air: Why indoor air quality is crucial for schools
Leave a CommentPicture this: A little girl sits in a classroom in her neighborhood elementary school. She’s trying to focus on the teacher’s lesson, but her asthma is acting up again. She’s wheezing and finding it hard to breathe. Her chest tightens. At some point, it gets to be too much. She has to leave school early […]

Microfiltration Matters
Leave a CommentH&V has developed innovative porous materials for over two centuries; microfiltration products for liquid filtration have been used by customers for over two decades: Wetlaid cellulose materials are used for semiconductor process water, electroplating, electrical discharge machining, and many other microfiltration processes. Wetlaid glass products are used in many analytical and test kit activities overseen […]

Celebrate Earth Day With NanoWave® Filter Media
Leave a CommentWhile Earth Day is an important time to celebrate nature and the beauty of the outdoors, at Hollingsworth & Vose, we are doing our part to make the air quality in buildings safer through filtration solutions that reduce energy consumption and minimize carbon emissions as compared to standard filtration in the market. With energy consumption […]

Hollingsworth and Vose Continues Its Pursuit of a Cleaner, Healthier, More Sustainable World
Leave a CommentHollingsworth & Vose has an innovative and insightful outlook that helped to grow the company from a family business established over 200 years ago to a global leader in the advanced filtration industry today. Our company’s primary goal is to develop sustainable filtration solutions for a cleaner, healthier world. Josh Ayer is the CEO of […]
Hollingsworth & Vose Continues to Expand Their Global Presence in India
Leave a CommentAs a trusted supplier of energy and filtration solutions, Hollingsworth & Vose is dedicated to serving customers around the world. With 13 production facilities strategically located in North America, Asia, and Europe, H&V has a global footprint that is positioned to meet the needs of regional customers. H&V’s international presence, and vertically integrated capabilities producing […]

How Does a Coalescer Filter Work?
Leave a CommentBasics of Coalescing Filtration Coalescer filtration is used to separate liquid droplets and aerosols from both liquid and gas media. Coalescer filter elements are manufactured from specialized materials, including borosilicate micro-fiberglass, synthetic fibers, and thermoplastic resins. Each coalescer filter material is tailored to the particular needs of the application, with a variety of filtration grades […]

Microfiltration vs. Ultrafiltration: What’s the Difference?
Leave a CommentMicrofiltration and ultrafiltration are separation methods that rely on filter media with tiny pores to remove solids from a liquid stream. Factors such as the pore size and contaminants present affect the degree of purification. Microfiltration and ultrafiltration strip solids from liquid streams until they are sent to more intensive treatment processes in a variety […]

The Importance of Microfiltration Membranes in Water Treatment
Leave a CommentWater cleanliness and filtration standards are rising in many parts of the world. That means municipal water treatment facilities and commercial water purification systems need to be increasingly advanced to keep up. One of the most important tools in an advanced filtration system is a microfiltration membrane. Microfiltration membranes use physical separation processes to remove […]

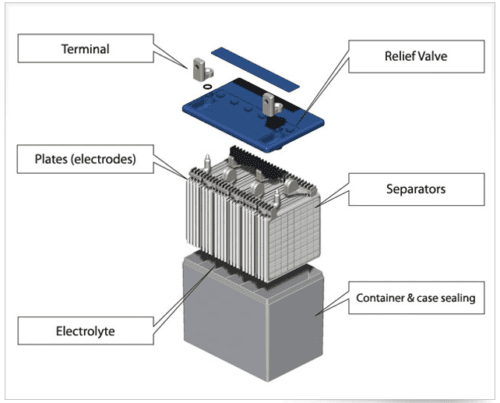
An Introduction to Battery Separators
Leave a CommentBatteries consist of the following basic components: an anode, a cathode, and an electrolyte. However, another key part that is often overlooked is the battery separator. This component is vital to the functionality and performance of batteries. Below, we outline what it is, its origins, and its applications. What Is a Battery Separator? A battery […]




